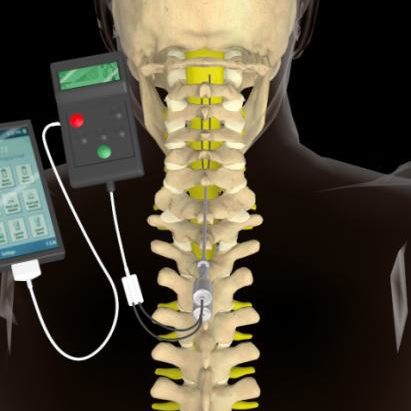
Do not use short-wave diathermy, microwave diathermy, or therapeutic ultrasound diathermy (all now referred to as diathermy) on patients implanted with a neurostimulation system. Patients without an MR Conditional neurostimulation system should not be subjected to MRI because the electromagnetic field generated by an MRI may damage the device electronics and induce voltage through the lead that could jolt or shock the patient.ĭiathermy therapy. Physicians should also discuss any risks of MRI with patients. A physician can help determine if a patient is eligible to receive an MRI scan by following the requirements provided by Abbott Medical. Scan if all the requirements for the implanted components and for scanning are met. Some patients may be implanted with the components that make up a Magnetic Resonance (MR) Conditional system, which allows them to receive an MRI Neurostimulation should not be used on patients who are poor surgical risks or patients with multiple illnesses or active general infections. The following warnings apply to this neurostimulation system. For more information about MR Conditional products, visit the Abbott Medical product information page at Some models of this system are Magnetic Resonance (MR) Conditional, and patients with these devices may be scanned safely with magnetic resonance imaging (MRI) when the conditions for safe scanning are met.įor more information about MR Conditional neurostimulation components and systems, including equipment settings, scanning procedures, and a complete listing of conditionally approved components, refer to the MRI procedures clinician's manual for neurostimulation systems (available online at This system is contraindicated for patients who are unable to operate the system or who have failed to receive effective pain relief during trial stimulation.  
The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Read this section to gather important prescription and safety information. Passive Recharge Burst Spinal Cord Stimulation Provides Sustainable Improvements in Pain and Psychosocial Function: 2-year Results From the TRIUMPH Study. Spine, 47(7), 548–556. G., Fraunberg, V., Blomme, B., & Capobianco, R. Jude Medical's Prodigy Chronic Pain System with Burst Technology. 2021.ĭe Ridder D, Vanneste S, Plazier M, Vancamp T. Proclaim™ Implantable Pulse Generator Clinician’s Manual. History of electrical neuromodulation for chronic pain. Pain Med. 2006 7(Supp 1):S7–13. Talk to your doctor about complications related to the procedure and/or device, which include infection, swelling, bruising, undesirable changes in stimulation and loss of strength or use in an affected limb or muscle group (e.g., paralysis). Only your doctor or pain specialist can determine if SCS or DRG may work for you. Neurostimulation is not right for everyone.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
